1 minute (overview).
Key Considerations for the Development of Lipid Nanoparticles for RNA Delivery
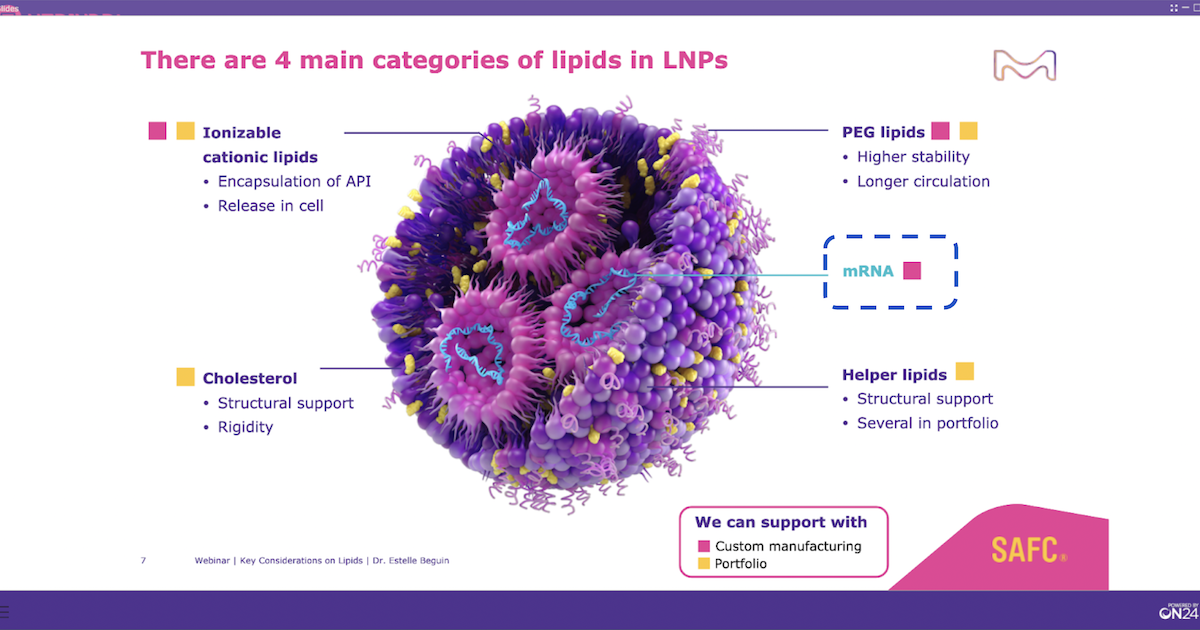
The success of mRNA-based vaccines for COVID-19 lays the path to accelerate the development of this modality for many other diseases. Key decisions must be made early in the drug development process for all aspects: from mRNA manufacturing to sourcing of critical components such as lipid excipients for the final formulation. Doing a deep dive into the critical aspects of these lipids, this webinar aims to highlight that considerations such as excipient quality can significantly impact the performance, reproducibility, costs, and regulatory timelines of the final drug product.
In this webinar, you will discover:
- How we got to the lipid nanoparticles we have today
- Key considerations for their formulation
- What’s important to keep in mind when choosing lipid excipients for the development of your drug product
Speaker

Estelle Beguin, Ph.D.
Merck
Strategic Marketing Manager – Synthetic Lipids
Estelle has experience working as a formulation and process engineer on drug delivery programs for several biotech companies in Boston. She has joined Merck as a global strategic marketing manager for synthetic lipids. In this role, she is responsible for managing the lipids portfolio and custom manufacturing businesses supporting top industry players and young start-ups in the fields of RNA delivery and vaccines. She holds a Ph.D. in Engineering Science from the University of Oxford, the U.K. where she worked on developing ultrasound-responsive lipid-based drug delivery systems.